Tulsa, OK
Tulsa, OK

Tulsa
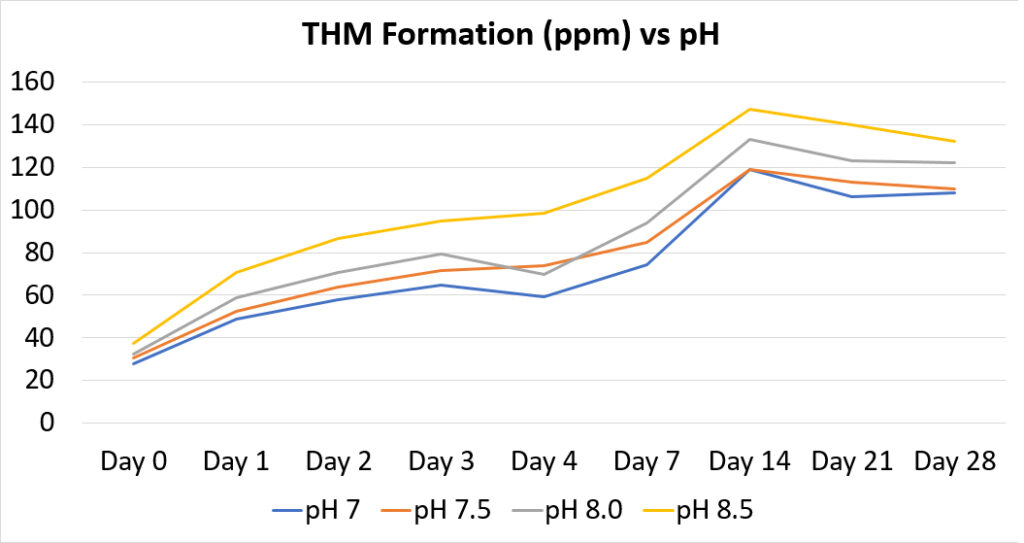
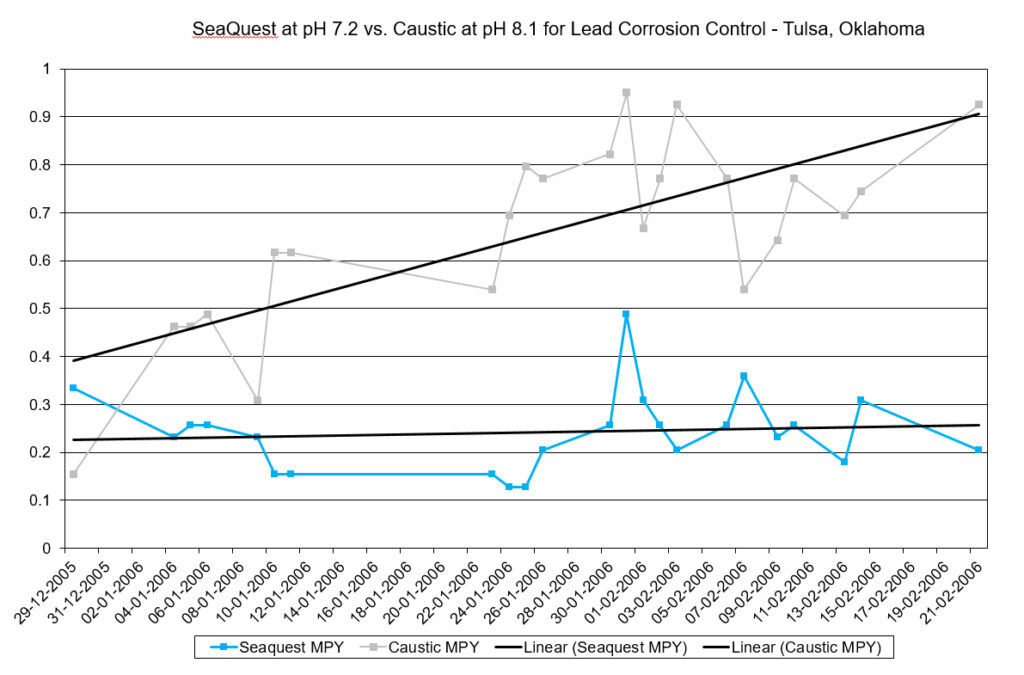
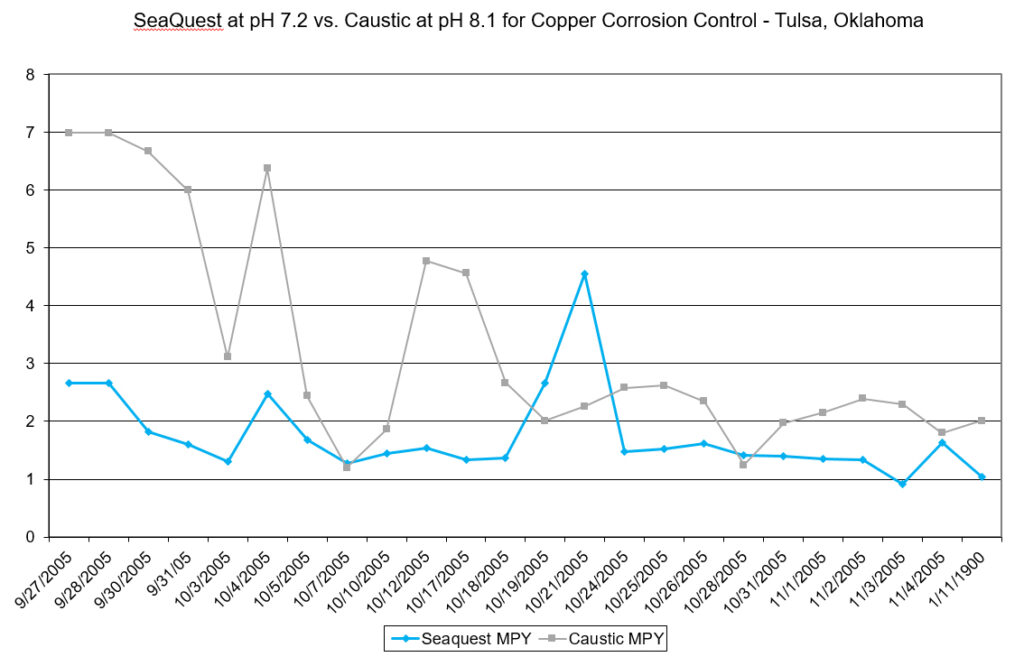
The City of Tulsa, OK had a problem with THM formation. Because they operated their system at a pH of 8.3, excess sodium hypochlorite was needed to produce free chlorine residuals. They confirmed through a study that a reduction in pH would result in less THMs. However, they feared a reduction in pH would result in higher levels of corrosion. Using SeaQuest:
- Lead corrosion rates were reduced by 65%
- Copper corrosion rates were reduced by 47%
- pH was reduced from 8.1 to 7.2
As a result, the tests showed the system could be operated at neutral pH to reduce disinfection byproducts (such as THMs) and actually have better corrosion control using SeaQuest.
Dave is the Vice President and General Manager of AquaSmart, and a Chemical Engineer.
